 25249) and transferred to Thermo Scientific PVDF Transfer Membrane (Part No. Samples were loaded onto 4-20% Thermo Scientific Precise Tris-Glycine Gels (Part No. Western blotsįifty micrograms of HeLa lysate was serially diluted (2-fold in each lane) in 4X LDS Sample Buffer (Product # 84788) supplemented with DTT at a final concentration of 50mM. For publication, the white level contrast was adjusted on the images acquired with the myECL Imager to display low-intensity pixels. For analysis, a manual region of equal size was placed over each of the eight reference plate light spots and the density (pixel intensity/region area) was plotted against the observed RLU (Relative Light Units) based on the manufacturer’s Certificate of Analysis. Image analysis was performed using the Thermo Scientific myImageAnalysis Software. To create digital images of the film for analysis, each film was scanned with an Epson™ 4990 Photo Scanner as a 300 PPI (pixels/inch), 16-bit grayscale TIFF image to match the image output from the myECL Imager. The film was developed in a Konica Minolta™ SRX-101A Film Processor with developer reagents. Images were captured on film and on the myECL Imager using all five binning settings (1 × 1, 2 × 2, 3 × 3, 4 × 4, and 8 × 8) at exposure times of 10, 30, 60, 120 and 300 seconds. The reference plate contains eight lights (spots) of varying, known signal intensities and is typically used for luminometer calibration. A qualitative assessment of the western blot examples shows that the myECL Imager detects an additional band of the HeLa lysate dilution series when compared to film (Figure 3).Ī Luminometer Reference Microplate (Harta Instruments, #RM-168) was used to demonstrate the imaging speed and sensitivity of the myECL Imager in comparison to standard X-ray film (Thermo Scientific CL-XPosure Film, Part No. Densitometry of these images shows a 1000-fold dynamic range (maximal signal intensity/minimal signal intensity) in the myECL Imager compared to a 1.5-fold dynamic range for the film, where dynamic range is calculated from the density of spot 1 divided by the density of spot 8 (Figure 2 Table 1). In contrast, only five of the eight spots are visible on a 300-second exposure to film. Seven of the eight reference plate spots are visible and distinguishable above background on the 300-second exposure acquired by the myECL Imager (Figure 2). Images acquired on the myECL Imager are more sensitive than film. Greater sensitivity and dynamic range compared with X-ray film (Developed films were then scanned at high resolution to produce digitized versions for analysis.) Finally we analyzed the images using the Thermo Scientific myImageAnalysis Software. We captured signals digitally using the myECL Imager and by exposure to X-ray film. To compare CCD camera and X-ray film imaging, we examined signals produced by a NIST-traceable Luminometer Reference Microplate (Harta Instruments, #RM-168) and by our own chemiluminescent Western blots containing serially diluted samples spanning the detectable range for imagers and film. Binning increases the size of each pixel, which effectively increases the amount of light collected in the pixel area. To enhance detection sensitivity further, the pixels, or light capturing units of the CCD chip, can be combined or binned. High-performance CCD cameras cool the silicon chip to sub-zero temperatures to reduce dark current, which produces background noise. Recent improvements in CCD technology have enabled the development and commercialization of sensitive, cooled-CCD cameras with higher light-capturing performance than film. 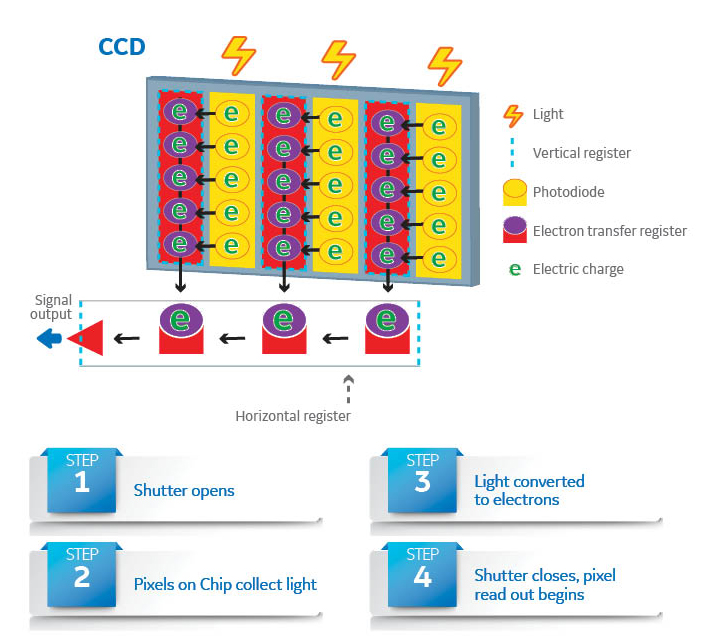
Imagers with early generation CCD cameras designed for low-light applications were not capable of matching the speed or sensitivity of X-ray film. Charge-coupled device (CCD) cameras use a light-sensitive silicon chip that converts photons into digital signals. Scanning of the film with a basic document scanner and the use of image analysis software has been used to extract some quantitative information however, densitometry from the scanned X-ray film image can be challenging (Ref.1) and chemiluminescent western blot analysis is generally regarded as not being quantitative.Īn alternative method to X-ray film for capturing luminescent signals from western blots is to use a cooled-CCD camera. Relative differences in protein amounts can be assessed by the signal intensity as it typically correlates to the protein amount in the blot. Western blot analysis has been used as a qualitative assessment of the presence of a specific protein of interest in a sample with a target-specific antibody. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
